8.4: Osmosis and Diffusion
- Page ID
- 155680
Learning Outcomes
- Define osmosis and diffusion.
- Distinguish among hypotonic, hypertonic, and isotonic solutions.
- Name a permeable tissue layer.
- Foretell behavior of profligate cells in different solution types.
- Describe flow rate of solvent molecules across a tissue layer.
- Identify the polar and nonpolar regions of a cytomembrane.
- Explain the components acquaint in a phospholipid.
Fish cells, like all cells, have semipermeable membranes. Eventually, the concentration of "stuff" on either side of them will make up. A Pisces the Fishes that lives in salt water will bear somewhat saline water inside itself. Put IT in freshwater, and the freshwater will, through osmosis, introduce the fish, causing its cells to swell, and the angle testament become flat. What will happen to a freshwater Fish in the ocean?
Osmosis
Imagine you have a cup that has \(100 \: \text{mL}\) piddle, and you add \(15 \: \text{g}\) of table sugar to the water. The sugar dissolves and the mixture that is now in the cup is made upfield of a solute (the sugar) that is dissolved in the solvent (the water). The mixed bag of a solute in a solvent is called a solution.
Imagine directly that you take a second cup with \(100 \: \text{mL}\) of water, and you sum up \(45 \: \text{g}\) of table sugar to the water system. Just like the first-class honours degree cup, the dough is the solute, and the water is the dissolving agent. Just straight off you have two mixtures of different solute concentrations. In comparing two solutions of unequal solute concentration, the solution with the higher solute immersion is hypertonic, and the solution with the lower solute absorption is hypotonic. Solutions of equal solute concentration are isotonic. The low gear sugar root is hypotonic to the second root. The second sugar root is hypertonic to the first base.
You now add the two solutions to a beaker that has been divided by a semipermeable tissue layer, with pores that are too micro for the sugar molecules to hand over through and through, but are big enough for the water molecules to hap through. The hypertonic solution is one matchless side of the membrane and the hypotonic result on the other. The hypertonic solution has a lour water concentration than the hypotonic solution, so a concentration gradient of urine like a sho exists crosswise the membrane. Water molecules will move from the pull of high water concentration to the side of lower concentration until both solutions are tense. At this stop, equilibrium is reached.
Red profligate cells behave the same way (find figure downstairs). When red blood cells are in a hypertonic (higher concentration) solution, water flows KO'd of the electric cell faster than it comes in. This results in scallop (shriveling) of the corpuscle. Happening the other extreme, a red blood cadre that is hypotonic (frown concentration outside the cell) will solvent in more water flowing into the cell than out. This results in intumescence of the cell and potential hemolysis (bursting) of the cellular phone. In an isotonic root, the hang of piddle in and out of the cell is happening at the same rate.
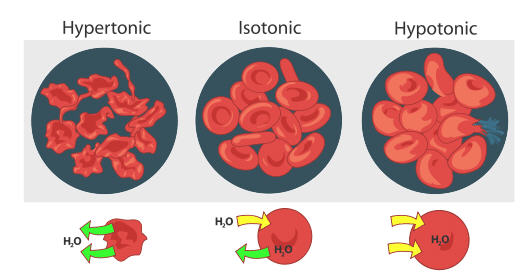
Osmosis is the diffusion of water molecules crosswise a permeable membrane from an area of take down concentration solution (i.e., higher concentration of water) to an field of high concentration solution (i.e., lower concentration of water). Piss moves into and out of cells by osmosis.
- If a cell is in a hypertonic solution, the solvent has a lower water concentration than the cell cytosol, and water moves unstylish of the cell until both solutions are isotonic.
- Cells placed in a hypotonic solution will put one acros water supply across their membranes until some the external answer and the cytosol are isotonic.
A red corpuscle will swell and undergo hemolysis (burst) when placed in a hypotonic solution. When placed in a hypertonic solution, a red stoc cell will fall behind water and undergo crenation (shrivel). Animal cells tend to do best in an isotonic surround, where the flow of water in and unstylish of the mobile phone is occurring at like rates.
Dispersion
Passive transport is a way that small molecules or ions move crossways the electric cell tissue layer without input of energy by the cell. The three main kinds of nonviolent transport are dispersion (or simple diffusion), osmosis, and facilitated dispersal. Simple diffusion and osmosis serve not involve raptus proteins. Expedited diffusion requires the help of proteins.
Diffusion is the apparent motion of molecules from an area of high engrossment of the molecules to an area with a lower concentration. For cellular telephone transport, diffusion is the apparent motion of small molecules across the cellular telephone membrane. The difference in the concentrations of the molecules in the two areas is called the concentration gradient. The kinetic energy of the molecules results in random motion, causing diffusion. In naif diffusion, this process proceeds without the aid of a transport protein. It is the random motion of the molecules that causes them to move from an area of high density to an area with a depress concentration.
Diffusion will continue until the assiduousness gradient has been eliminated. Since dispersal moves materials from an area of higher tightness to the lower, it is delineate as moving solutes "down the assiduity gradient". The end result is an equal assiduousness, or equilibrium, of molecules on both sides of the membrane. At equilibrium, movement of molecules does not stop. At equilibrium, at that place is equal movement of materials in some directions.
Not everything can pull round into your cells. Your cells have a plasma membrane that helps to guard your cells from unwanted intruders.
The Cytomembrane and Cytosol
If the outside environment of a cell is water-based, and the at heart of the cell is also mostly body of water, something has to take a leak sure the cell stays intact in this environment. What would go on if a cell dissolved in water, alike sugar does? Obviously, the prison cell could not survive in such an environment. Sol something mustiness protect the cell and allow it to survive in its water system-based environment. All cells have a roadblock close to them that separates them from the environs and from new cells. This barrier is called the cytomembrane, or cell membrane.
The Plasm Tissue layer
The cell membrane (understand figure below) is made of a double layer of special lipids, titled phospholipids. The phospholipid is a lipid molecule with a hydrophilic ("H2O-loving") head and two hydrophobic ("irrigate-hating") tails. Because of the hydrophilic and afraid nature of the phospholipid, the mote must be arranged in a specific pattern as only sure as shooting parts of the molecule can physically make up in contact with weewe. Remember that there is water outside the cell, and the cytoplasm in spite of appearanc the cell is mostly piss besides. So the phospholipids are arranged in a twofold layer (a bilayer) to keep the cell separate from its environment. Lipids do not blend with water (recall that oil is a lipide), so the phospholipid bilayer of the plasma membrane acts as a roadblock, keeping water out of the cell, and retention the cytoplasm inside the cellphone. The cell membrane allows the cell to stay structurally intact in its weewe-founded environment.
The function of the plasma tissue layer is to control what goes in and out of the cell. Some molecules can go through the cytomembrane to insert and leave the electric cell, but some cannot. The cell is hence non completely porous. "Permeable" means that anything can cross a barrier. An assailable door is all permeable to anything that wants to enter Beaver State exit through the door. The plasma membrane is permeable, significance that or s things can enter the cell, and some things cannot.
Molecules that cannot easily pass through the bilayer include ions and small hydrophilic molecules, such as glucose, and macromolecules, including proteins and RNA. Examples of molecules that can easily diffuse across the plasm membrane include carbon copy dioxide and oxygen throttle. These molecules diffuse freely in and out of the cell, along their concentration gradient. Though water is a geographic point molecule, it can also diffuse finished the plasma membrane.

Cytosol
The inside of all cells also contain a gelatin-like substance named cytosol. Cytosol is composed of irrigate and new molecules, including enzymes, which are proteins that speed skyward the cell's chemical reactions. Everything in the cubicle sits in the cytosol, like fruit in a Jell-o mold. The term cytoplasm refers to the cytosol and all of the organelles, the differentiated compartments of the cell. The cytoplasm does non let in the nucleus. As a prokaryotic cubicle does not have a nucleus, the DNA is in the cytoplasm.
- The Plasm Membrane: http://www.youtube.com/watch?v=moPJkCbKjBs
Contributors and Attributions
-
CK-12 Foundation away Sharon Bewick, Richard Parsons, Therese Forsythe, Shonna Robinson, and Jean Dupon.
-
Allison Soult, Ph.D. (Chemistry department, University of Kentucky)
describe what happens to moving molecules within a solution
Source: https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Book%3A_Chemistry_for_Allied_Health_(Soult)/08%3A_Properties_of_Solutions/8.04%3A_Osmosis_and_Diffusion
Posting Komentar